The Centre of Excellence in Cellular Immunotherapy Development Program provides access to a full spectrum of specialised cellular immunotherapy preclinical know-how, product manufacturing development and clinical trial capabilities.
The Development Program aims to expand the offering of ground-breaking cellular immunotherapy clinical trials that are available to Australians with cancer. It acts as an IP accelerator program, designed to speed up the process of getting new, cutting-edge therapies into early-phase clinical trials.
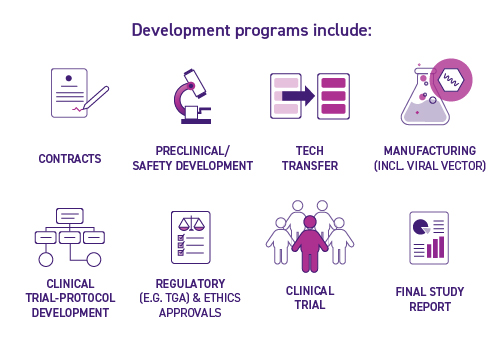
The Development Program also focuses on collaboratively developing new technologies for proof-of-concept clinical trials.
This may include:
- Preclinical, manufacturing and clinical development activities that continue where discovery research ends
- Provision for CAR-T or other cellular immunotherapy doses by the Centre’s GMP manufacturing partners, Cell Therapies Pty. Ltd.
- Supply of clinical/GMP grade materials including vector
- Necessary regulatory and governance expertise for TGA, HREC and associated governance approvals
- Peter Mac sponsored clinical trial with a Peter Mac Cancer Centre clinician as a Principal Investigator (PI)

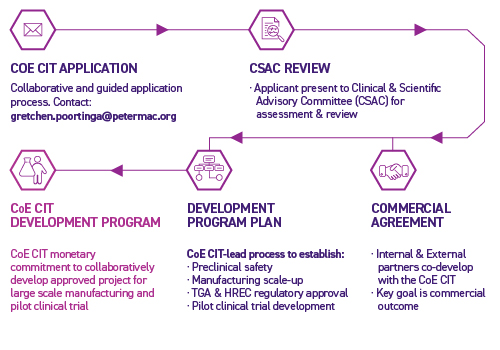
Centre of Excellence in Cellular Immunotherapy Pilot Clinical Trial Development Program Application Process
The Development Program application assessment process is overseen by the Centre of Excellence in Cellular Immunotherapy Clinical and Scientific Advisory Committee (CSAC). This Committee is made up of national and international experts in leading-edge cellular immunotherapy research and its clinical application, along with expertise in manufacturing development and commercialisation.
This Committee is responsible for choosing the most promising project applications based on selection criteria, which include aspects of:
- strong scientific rationale
- innovative concept, avoiding replica studies
- clinical feasibility
- commercial viability with a path to potential commercialisation
Our team invites discussions around projects prior to application. Please contact us for more information.
The Centre of Excellence in Cellular Immunotherapy invites discussions around projects prior to application.
Centre of Excellence in Cellular Immunotherapy application
Download the Centre of Excellence in Cellular Immunotherapy application
Frequently asked questions about the Pilot Clinical Trial Development Program
Contact
For more information contact
